The science of concussive symptoms
The exact mechanisms behind concussive symptoms remain unclear. Research suggests that trauma stretches nerve cell membranes and fibers (axons), triggering a neurometabolic cascade that leads to neuroinflammation and potential injury or cell death.
A common misconception is that concussions require loss of consciousness, which occurs only in a minority of cases. While severe neurological symptoms like post-traumatic amnesia, disorientation, epilepsy, or paralysis can result, mild traumatic brain injuries often present subtler signs, such as headaches, difficulty concentrating, a sense of mental fog or pressure, fatigue, irritability, and drowsiness.
Sports evaluation tools, such as the SCAT6 (Sport Concussion Assessment Tool) designed for individuals aged 13 and older, are used within 72 hours of injury, while the SCOAT (Sport Concussion Office Assessment Tool) assesses symptoms after three days. These tools help identify non-specific symptoms, track recovery, and grade concussions as mild, moderate, or severe, depending on the classification system.15
Treatment focuses on managing symptoms through rest and avoiding further head impacts. However, early, light activity is often recommended over complete rest. Ensuring full symptom resolution before returning to high-risk activities is critical to avoid “second impact syndrome,” a rare but life-threatening condition.
Post-concussion syndrome and persistent symptoms
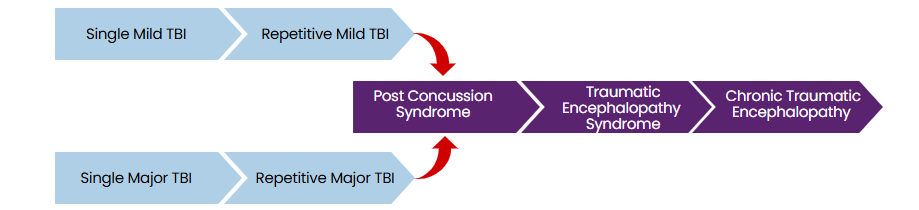
When concussive symptoms persist for four weeks to three months, the condition is termed post-concussion syndrome (PCS) or persistent post-concussive symptoms (PPCS). These terms, used interchangeably, can arise from repetitive mild TBIs (rmTBI), as well as moderate or severe TBIs.
The development of PPCS likely involves a complex interplay of biological factors, such as diffuse axonal injury, neuroinflammation, and altered cerebral blood flow, alongside psychological, socio-demographic, and personality factors. Incidence rates vary widely across studies due to differing diagnostic criteria, and controversy persists over whether PCS can be fully attributed to mild TBI. Subjective symptoms, which include headaches, fatigue, and mood changes, are non-specific and may predate the injury, complicating definitive diagnosis. Studies have even observed similar symptoms in healthy populations, non-head trauma patients, and individuals with chronic pain.4,5,7
An estimated 15% of individuals with mTBI develop PPCS, although some studies suggest rates as high as 25%, depending on the criteria used.8,9
Treating PCS
Managing PCS is particularly challenging due to the complexity of symptoms and limited evidence for some treatments. Current approaches include:
Non-pharmacological interventions
- Cognitive rehabilitation: Helps improve cognitive deficits in attention, memory, and executive functioning.
- Psychotherapy such as cognitive behavioral therapy (CBT): Provides coping strategies for emotional symptoms like irritability, depression, and anxiety.
Pharmacological treatments
- Antidepressants and anti-anxiety medications: Used symptomatically to manage emotional and psychological symptoms.
- Stimulants: Sometimes prescribed to improve attention and concentration.
Rehabilitation therapies
- Vestibular and vision therapy: Addresses cervical soft tissue damage often associated with mTBI, although evidence of efficacy remains limited.
The Australian Sports Commission’s 2024 “Concussion and Brain Health” position statement highlights a physiotherapy-guided rehabilitation plan, focusing on five key areas – the autonomic system, cervical spine, vestibular function, vision, and cognition – to guide an athlete’s safe return to activity.13,14,15
Traumatic encephalopathy syndrome and chronic traumatic encephalopathy
The neuropathological hallmarks of chronic traumatic encephalopathy (CTE-NC) include hyperphosphorylated tau (pTau) protein and neurofibrillary tangles (NFTs) or pretangles in specific brain regions characteristic of the condition. These abnormalities cluster around small blood vessels in the cerebral cortex, leading to progressive and debilitating neurodegeneration.
While tau deposition is common in several neurodegenerative diseases, such as Alzheimer’s, and in normal aging, the pattern and distribution of tau and NFT pathology in CTE are distinct enough to be considered diagnostically definitive.
The mechanisms underlying CTE remain unclear, but repetitive mild traumatic brain injuries (rmTBI) are believed to cause abnormal phosphorylation of tau protein. This misfolded and cleaved protein aggregates into clumps, potentially promoting the accumulation of other aggregate-prone proteins. These pathological changes can be confirmed only through autopsy, as they are not detectable via imaging.
The pathological stages of CTE are illustrated below:
The term CTE-NC (neuropathological changes) refers specifically to the condition diagnosed postmortem. In contrast, traumatic encephalopathy syndrome (TES) describes the clinical signs and symptoms associated with CTE-NC in individuals who are still alive.
Both conditions share features such as impairments in higher executive functioning, including short-term memory loss, mood instability, depression, and difficulties with decision-making and multitasking. TES is also linked to later-life conditions such as dementia and Alzheimer’s disease, and movement disorders like Parkinson’s disease.16
Controversies surrounding CTE
The relationship between repetitive mild traumatic brain injuries (rmTBI) and chronic traumatic encephalopathy (CTE-NC) remains controversial, with ongoing debate over whether rmTBI causes CTE-NC or is correlated with it. CTE-NC is a condition that develops over decades, requiring evaluation of head trauma from the distant past. Each mTBI is typically brief, often ambiguous, and rarely quantified, making rigorous clinical studies difficult.12
Research on CTE-NC largely consists of case reports, retrospective studies, and post-mortem analyses, often with selection bias. Many diagnoses come from retired athletes who donated their brains to sports brain banks.
Environmental factors, such as substance use, genetic predisposition, mental health history, and education level, further complicate causation studies. A 2023 study of 636 cases from the Sydney Brain Bank confirmed a low prevalence of CTE-NC in the general population (0.8%), with only five cases identified -three involving a history of TBI and two without.12
The Concussion in Sport Group’s 2023 “Consensus Statement on Concussion in Sport” acknowledged the potential link between extensive repetitive head impacts, such as those experienced by some professional athletes, and the neuropathology of CTE-NC. This influential group’s concussion protocols are widely adopted across elite and community sports worldwide.17
Investigations for diagnosing concussion
With no specific test or biomarker available, concussion diagnosis and monitoring rely on identifying relevant symptoms and clinical signs.
However, advancements in biomarkers and imaging technologies offer promising new tools for diagnosing mild traumatic brain injuries.
Fluid biomarkers
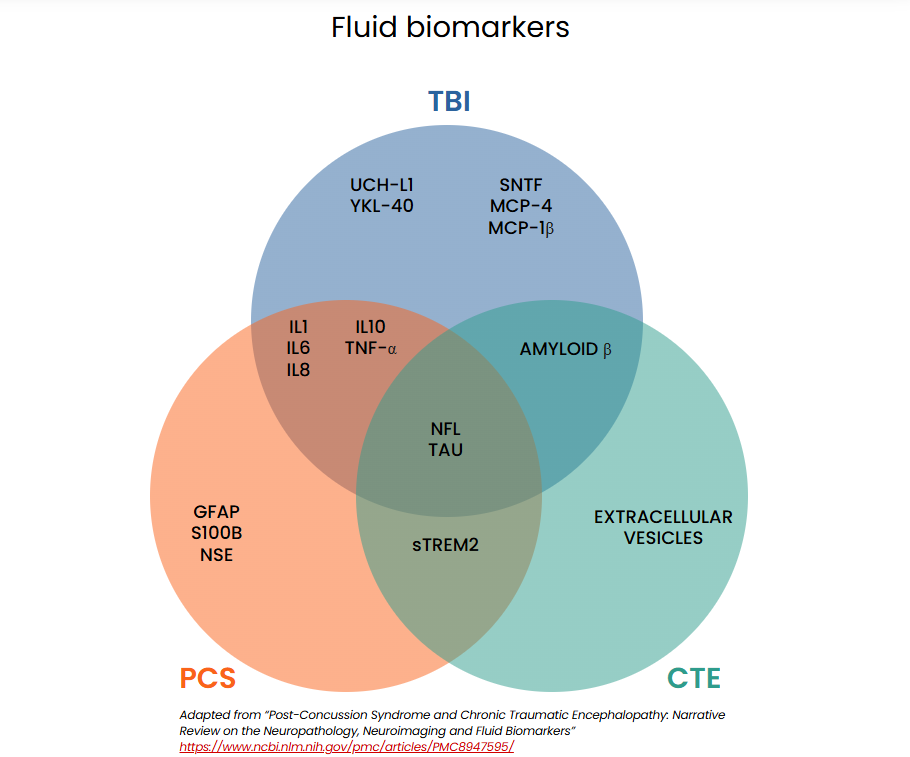
Brain injuries can cause proteins to leak from damaged cells into cerebrospinal fluid (CSF) or cross the blood-brain barrier into the bloodstream, where they can be measured. This evolving area of research shows promise for diagnosing mTBI. Biomarkers in blood, CSF, saliva, and urine are under investigation to help distinguish concussions from non-concussive head injuries, differentiate mild from severe intracranial injuries, and identify individuals at risk for prolonged symptoms.
The schematic below illustrates biomarkers currently being studied for diagnosing concussion-related conditions.15,18,19
FDA-approved biomarker testing
In February 2018, the US Food and Drug Administration (FDA) approved the first blood test for detecting intracranial injury following a mild traumatic brain injury. This test measures two proteins: ubiquitin C-terminal hydrolase-L1 (UCH-L1) and glial fibrillary acidic protein (GFAP), markers of neuronal and glial cell damage. Because each protein originates from different brain cell types, their combined measurement provides complementary insights into intracranial injury. The test was approved specifically for emergency care use in adults over 18 to reduce unnecessary CT scans in patients with mTBI. A negative result within 12 hours of injury is associated with the absence of acute intracranial lesions on CT imaging.
The test is used alongside clinical information, Glasgow Coma Scale (GCS) scores (13-15 for mTBI), and clinical imaging guidelines, such as the Canadian CT Head Rule.21,22
Limitations
- Not validated for individuals under 18 years old
- Detects intracranial bleeding rather than diagnosing concussions specifically
- Approved only for use in emergency settings and within 12 hours of injury
In April 2024, the FDA approved a portable version of the test that delivers lab-quality results within 15 minutes and can evaluate patients up to 24 hours after injury.24 The US Army has partnered with a manufacturer to deploy this proprietary TBI test in field settings, citing nearly 500,000 troops who experienced TBI during training or deployment from 2000 to 2023.25
While these advancements show promise, current biomarkers cannot determine when the brain has fully healed, making them insufficient for guiding return-to-work or return-to-sport decisions. Furthermore, the longer the delay between injury and testing, the less sensitive the results. Continued research into biomarkers holds significant potential for improving concussion diagnostics and care.
Specialized MRIs and other diagnostic tests
Imaging
Conventional CT and MRI scans are not effective for evaluating mTBIs or concussions, as these injuries often fall below the detection threshold or are too mild for individuals to seek imaging. While useful for ruling out structural damage and bleeding, these methods lack the sensitivity needed to diagnose mTBIs. Even with normal CT results, individuals can experience persistent symptoms for months or years following an mTBI.
Over the past decade, significant advances in imaging technology have improved the ability to detect milder TBI damage. However, the high cost and limited availability of these advanced modalities, especially in low- and middle-income countries, pose barriers to widespread adoption.
Specialized MRIs
- Diffusion tensor imaging (DTI): DTI detects microstructural white matter changes associated with diffuse axonal injuries, a critical finding in mTBI. By measuring the magnitude and direction of water diffusion in the brain, DTI provides a detailed view of neural pathways that standard MRIs cannot detect. This technique shows promise for earlier and more accurate diagnoses of mTBI-related damage.
- Functional MRIs (fMRI): Used in surgical planning for epilepsy and brain tumor removal, fMRI maps brain activity during specific tasks (e.g., motor, language, visual, or memory tasks). In concussions, fMRI reveals disruptions in brain networks, including the “default mode network,” which is most active during resting states. Similar disruptions are seen in conditions like Alzheimer’s disease and schizophrenia, although the exact reasons remain unknown.15
Other diagnostic tests
- Positron emission tomography (PET): PET scans measure areas of reduced brain metabolism to assess the severity of neural disruption. While studied for detecting abnormal tau tangles seen in CTE, current results are inconsistent, with low sensitivity and specificity, limiting their utility.
- Neuropsychological testing (NPT): NPT identifies cognitive and behavioral deficits following TBI. When combined with imaging modalities like DTI or fMRI, NPT may enhance the identification of affected brain regions, paving the way for more individualized treatment and rehabilitation strategies.11
Implications for the insurance industry
The growing recognition of concussion-related conditions like chronic traumatic encephalopathy (CTE) and persistent post-concussion symptoms presents unique challenges for insurers. With evolving medical research and increasing legal and regulatory scrutiny, the industry must adapt to manage these risks effectively. From underwriting models to claims management and policy design, insurers face significant opportunities and responsibilities to address the long-term implications of concussions.
- Risk assessment and underwriting – Underwriting must account for concussion-related risks, not only in high-contact sports but also across broader demographics. Individuals with a history of mTBI or rmTBI may face an increased likelihood of persistent symptoms or long-term conditions like CTE. Insurers may need to revise traditional underwriting models, incorporating evidence-based risk assessments and exclusions to mitigate potential long-term liabilities, particularly in disability insurance.
- Claims management – Persistent concussive symptoms and related conditions often lack a clear resolution timeline, requiring vigilance from claims teams. Training claims assessors to manage complex neurological claims and coordinate with healthcare providers is essential for timely interventions. Insurers must prepare for the possibility of extended claims as these conditions may progress over time.
- Policy design and coverage limits – Policy structures may need adjustments to balance adequate coverage for concussion-related injuries and long-term risks like CTE with sustainability. This applies both to high-risk groups, such as athletes, and to broader populations.
- Legal and regulatory considerations – The growing body of research and legal precedents surrounding concussion-related injuries, particularly in high-contact sports, poses challenges. Insurers may face disputes over coverage or exclusions and should stay informed about litigation trends and regulatory changes. If a definitive causal link between mTBI and CTE emerges, insurers may also need to consider occupational health and safety regulations, as such risks could be deemed preventable.
- Policyholder education and awareness – Insurers can contribute to concussion awareness by educating policyholders on prevention, early recognition of symptoms, and the importance of timely treatment. Resources could include safety guidelines for sports, post-TBI care recommendations, and advice on managing risks.
Navigating the uncertainty: The path forward for insurers
The challenge for insurers lies in identifying individuals at risk of long-term consequences from mild traumatic brain injuries (mTBIs) while distinguishing them from the majority who recover without complications. Predicting and assessing this risk remains complex, with current medical science offering limited tools to address the uncertainty.
Until advancements in medicine and science provide more definitive answers, insurers face a difficult balancing act: managing a complex clinical condition while mitigating financial exposure. To navigate this uncertainty, insurers must adapt underwriting practices, refine risk models, and ensure policies address emerging needs. Staying informed about the latest research and innovations is essential for building strategies that both support policyholders and maintain sustainability.
By addressing these challenges with a proactive and informed approach, insurers can better serve their policyholders while navigating the complexities of an ever-evolving healthcare landscape.
With thanks to Dr. John Mayhew for sharing his generous time and deep expertise on this topic.